1
REVOLUTION
ENGLISH
45MHz ROTATIONAL IMAGING CATHETER
CAUTION:
1. U.S. Federal Law restricts this device to sale by or on the order of a
physician.
2. Prior to use, read this entire Instructions For Use.
INTENDED USE:
The Revolution
®
catheter is intended for the intravascular ultrasound
examination of coronary arteries. Intravascular ultrasound imaging is indicated in
patients who are candidates for transluminal interventional procedures.
DESCRIPTION:
The Revolution 45MHz rotational IVUS imaging catheter consists of two main
assemblies; the imaging core and the catheter body. The catheter body
comprises of three sections; distal section w/ .014" compatible F/X port,
proximal section (single lumen), telescope section.
The distal section and proximal (single lumen) sections comprise the "working
length" of the catheter, the telescoping section remains outside of the guiding
catheter. The telescoping shaft (section) allows the imaging core to be advanced
and retracted for up to 150 mm of linear movement. The corresponding movement
of the transducer occurs from the proximal end of the guidewire exit port to the
proximal end of the window portion of the distal section.
The imaging core is composed of a hi-torque, flexible, rotating drive cable with a
distal outward looking 45MHz ultrasonic transducer. An electromechanical
connector interface at the proximal end makes the connection to the patient
interface module (PIM). The PIM-catheter interface consists of an integrated
mechanical drive assembly and electrical connection.
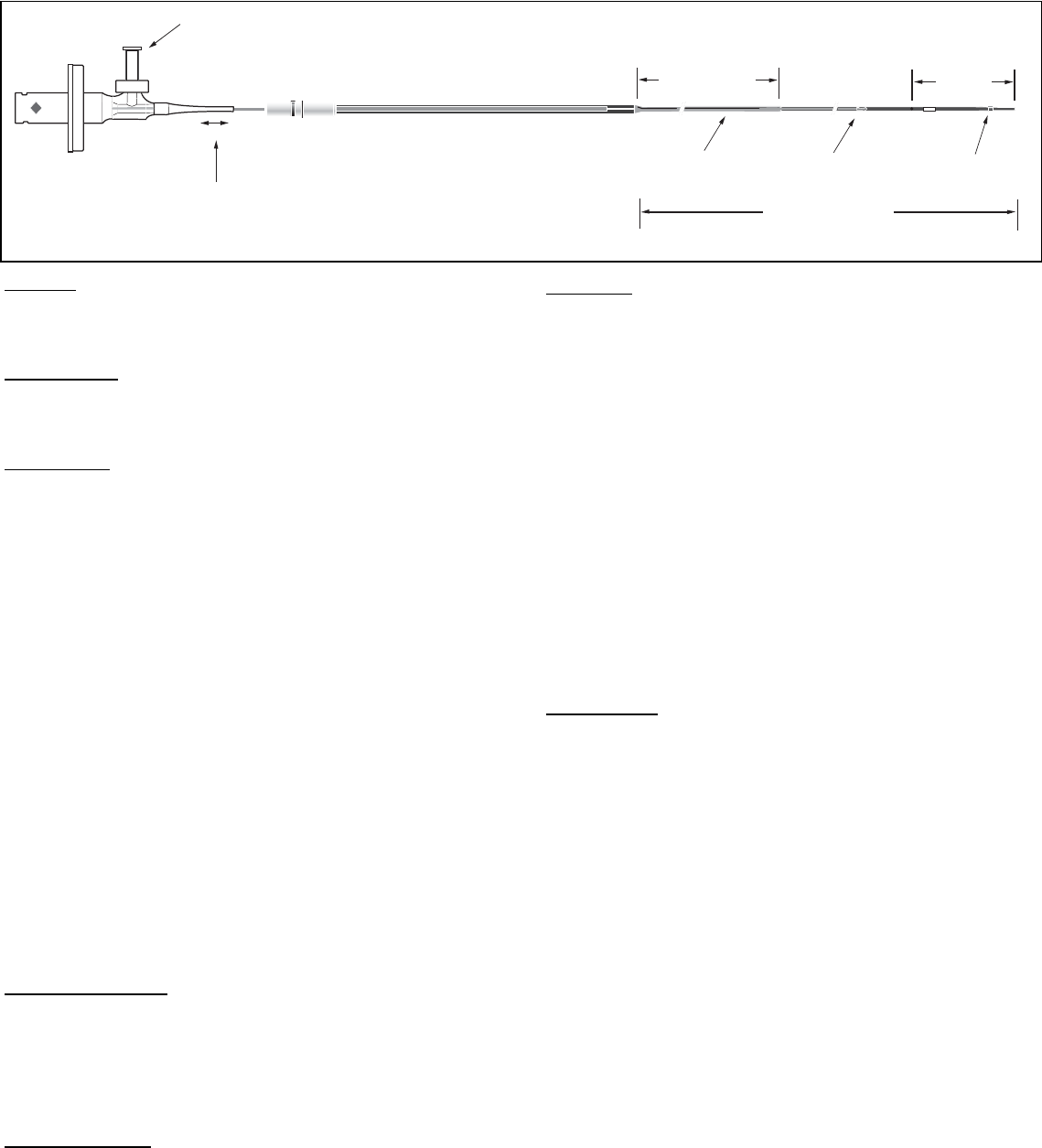
A flushing port with a one-way valve (Fig. 1) is used to displace the air initially
present within the catheter. The catheter must be flushed with heparinized
saline prior to use, as this provides the acoustic coupling media required for
ultrasonic imaging. The one-way valve helps retain saline in the catheter
during use.
The catheter body has a distal guidewire lumen with a proximal exit port
located 2 cm from the distal end (Fig 1). A radiopaque (RO) marker is
embedded in the catheter body at 0.5 cm from the tip. In addition, an insertion
depth indicator is located on the catheter body at 100 cm, corresponding to
femoral insertions.
The catheter is for use with the In-Vision Gold imaging system with software
V5.0 or higher or the Volcano s5 and Volcano s5i imaging system. Consult
your System Operator’s Manual.
CONTRAINDICATIONS:
This device is not currently indicated for use in cerebral or peripheral vessels.
Use of IVUS Imaging Catheters is contraindicated where introduction of any
catheter would constitute a threat to patient safety. Contraindications include:
bateremia or sepsis, major coagulation system abnormalities, patients
disqualified for CABG surgery, patients disqualified for PTCA, severe
hemodynamic instability or shock, patients diagnosed with coronary artery
spasm, and total occlusion.
ADVERSE EFFECTS:
Bleeding at the entry puncture site, injury to the vascular wall, thrombosis of
the vessel, and peripheral embolization has occurred with the use of
percutaneous intravascular catheter devices.
WARNINGS:
Use of the Revolution catheters is restricted to specialists who are familiar
with, and have been trained to perform, the procedures for which this device
is intended.
DO NOT advance the catheter if resistance is encountered. The catheter
should never be forcibly inserted into lumens narrower than the catheter
body or forced through a tight stenosis.
Care should be taken when utilizing devices that comprise a short monorail;
in such instances advancement of the device distal to a deployed stent can
result in exposure of the guidewire to the stent struts.
In instances where the device has crossed a deployed stent, care should be
taken when retracting the device to ensure that entanglement does not
occur. Fluoroscopy should be used to monitor guidewire position with the
respect to the imaging catheter and the stent; at no time should the imaging
catheter be retracted if there is evidence of guidewire prolapse or if
significant resistance to withdrawal is experienced. If either of these events
occur, advance the imaging catheter distal of the stent and then carefully
remove the whole system under the guidance of fluoroscopy.
Care should be taken when re-advancing a guide wire after stent deployment. A
guide wire may exit between stent struts when re-crossing a stent that is not fully
apposed to the vessel wall. Subsequent advancement of the catheter could
cause entanglement between the catheter and the stent. Care should be taken
to slowly remove the catheter from a stented vessel.
PRECAUTIONS:
The Revolution device is a delicate scientific instrument and should be treated
as such. Always observe the following precautions:
Contents supplied STERILE using an EtO (ethylene oxide) process. Do not
use if sterile barrier is damaged. If damage is found call your Volcano
Corporation representative.
To maintain optimal patient safety, inspect the product prior to use. Do not
use if saline leaks from any location other than the vent port in the monorail
section.
For single use only. Do not re-use, reprocess or re-sterilize. Re-use,
reprocessing or re-sterilization may compromise the structural integrity of
the device and/or lead to device failure which in turn may result in patient
injury, illness or death.
Re-use, reprocessing or re-sterilization may also create a risk of
contamination of the device and/or cause patient infection or cross-infection,
including, but not limited to the transmission of infectious disease(s) from
one patient to another. Contamination of the device may lead to injury,
illness or death of the patients.
The catheter has no user serviceable parts. Do not attempt to repair or to
alter any component of the catheter assembly.
Do not attempt to connect the catheter to electronic equipment other than
the designated systems.
Never attempt to attach or detach the catheter while the PIM motor is
running. To do so may damage the connector.
Avoid any sharp bends, pinching, or crushing of the catheter.
Do not kink or sharply bend the catheter at any time. This can cause drive
cable failure. An insertion angle greater than 45º is considered excessive.
Turn the PIM "OFF" before withdrawing the imaging catheter.
Fig. 1
One Way Luer
Telescoping Section
Pullback Length
150 mm
Proximal Shaft
109 cm
Usable Length
135 cm
Monorail
23mm
RO
Marker
3.5F
3.2F


















